Individualizing drug therapy based on pharmacogenomics (PGx) is considered by many academic researchers and clinical experts as the “next significant domain of clinical practice” (see the American College of Clinical Pharmacology’s very informative web-based tutorial, “The Future of Medicine: Pharmacogenomics”, http://user.accp1.org/index_new.html).
Drugs that are currently recommended for PGx testing by the FDA include specific medications that fall into these specialties:
- Oncology
- Cardiovascular, including warfarin and Plavix®, as well as the new generation of anticoagulants
- Neuropharmacology
In a study conducted by the large Pharmacy Benefits Management (PBM) company Medco Health Solutions (MEDCO) and the American Medical Association, based on responses from more than 10,000 physicians nationwide, less than 25% had any type of education in PGx, and only 10% of physicians felt that they have the adequate skills and training necessary to use PGx testing for their patients.
There is almost universal agreement that PGx gene test results will have to be provided to the clinician through the use of Clinical Decision Support Systems, integrated into the Electronic Health Record (EHR), through web-based portals such as www.warfarindosing.org, and/or software such as GeneMedRx® (Genelex). In the EHR vendor community, McKesson has been a pioneer in the integration of genomic and PGx functionality into their product offerings.
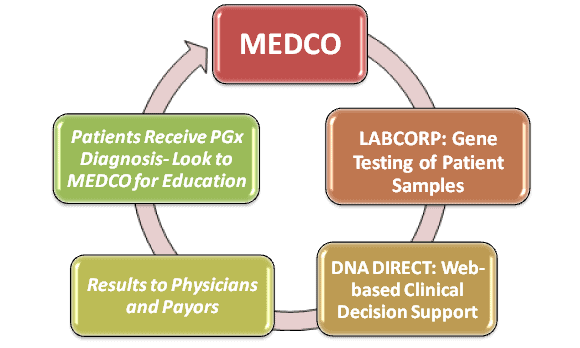
However, there is a compelling argument that pharmacists should play a decisive role in PGx. Large PBMs have positioned themselves as “gate-keepers” for PGx decisions. For example, MEDCO, that serves 60 million patients, has:
- Recently established specialty pharmacies staffed by “specialty pharmacists’ in domains such as oncology, cardiovascular, and psychiatry/neurology.
- Acquired DNA Direct, Inc., a leader in providing guidance and web-based decision support in genomic medicine to patients, providers, payors and employees.
- Started a head-to-head study of Plavix® (clopidogrel) and Effient® (prasugrel) that measures how the effectiveness of these drugs in heart patients is impacted by their genetic make-up. The study will examine whether the 70 to 75 percent of patients who are “extensive metabolizers” of clopidogrel – because they were born with a normally functioning version of the CYP2C19 gene – have comparable outcomes to those patients taking prasugrel, a newer, higher cost drug with metabolism less dependent on genetic variations.
The FDA is very supportive of the potential for PBMs to play the key role as PGx decision-makers.
In hospital systems, physicians typically work closely with pharmacists, so having PBM’s provide them with PGx decisions may make sense. However, will these solutions be provided through EHRs, or delivered by subsidiaries such as DNA Direct as an extension of laboratory gene testing? This could be an interesting time to gauge which IT solutions will have the greatest impact in this emerging medical domain.
Hypothetical business model:



Share Your Thoughts
You must be logged in to post a comment.